 For carbon, we multiply its molar mass of 12.0107 grams per mol by. Let's practice determining the molar mass of some common substances. Chemistry is a physical science, and it is the study of the properties of and interactions between matter and energy. The mass of a mole of atoms of an element is called the atomic mass and is given in the periodic table. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. mole of carbon atoms and two moles of oxygen atoms. Now we interpret the coefficients as referring to molar amounts, not individual molecules. The periodic table lists the molar mass of each element. Simulations - Discover a new way of learning Physics using Real World Simulations. 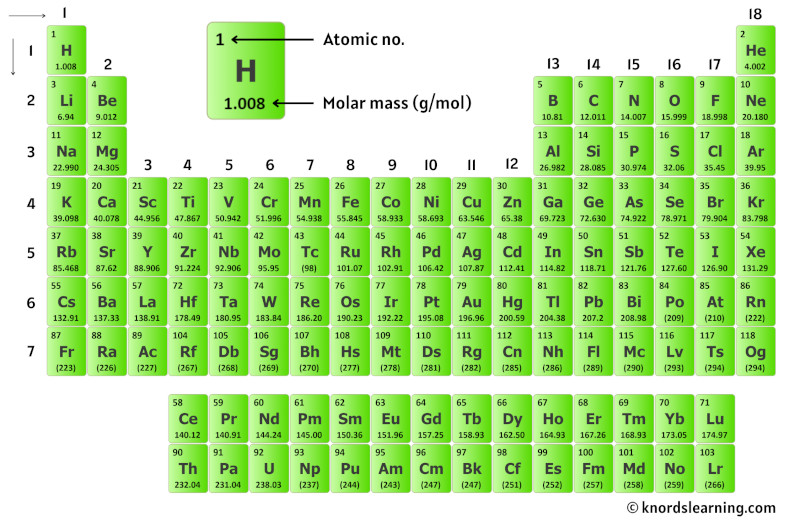
We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, Mu 1 × 103 kgmol1. Instead of using either Hydrogen, or Oxygen as the standard, the isotope of Carbon with 6 protons and 6 neutrons in its nucleus (Carbon-12) was given a mass of. But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as 
To find the mole fraction and percentage of each element in C3H8, divide each total from step 3 by the total molar mass found in step 4: Mole Fraction. 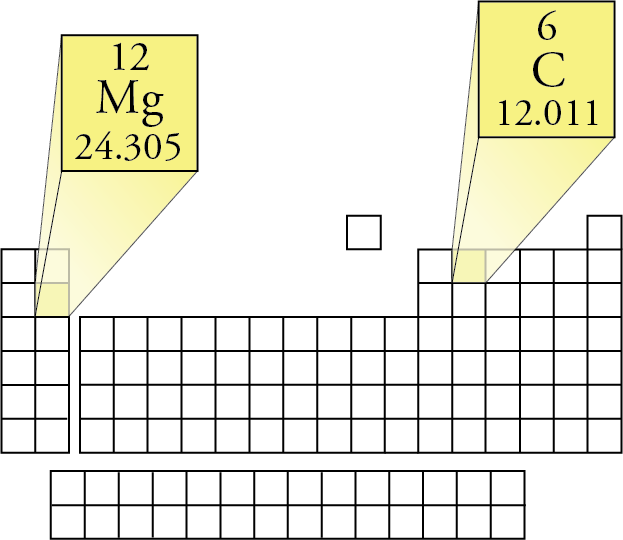
These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced. Finally, add together the total mass of each element to get the molar mass of C3H8: 36.0321 g/mol + 8.06352 g/mol 44.09562 g/mol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
